What do you think of this article?
By Dr. David Woo - Published on October 23, 2023
Last updated on November 8, 2024
Updated on September 18, 2024
Major depressive disorder (MDD) affects more than 16 million adults in the United States (1). Standard therapies for major depression include antidepressant medications and talk therapy. However, many patients don’t find relief from depressive symptoms with traditional treatments such as pharmaceuticals—this is referred to as treatment-resistant depression (TRD). The acknowledgment that treatment-resistant depression is a reality for many patients has led scientists to explore other treatment options besides the standard use of medication and psychotherapy. Transcranial magnetic stimulation (TMS) and deep brain stimulation (DBS) are two different therapies used to treat TRD. If you feel that your depression medications aren’t working, then either TMS or DBS may be right for you. However, there are major differences between the two therapies that patients should be aware of. This article compares deep brain stimulation vs. transcranial magnetic stimulation to help you make an informed decision.
Jump to:
- What Is Deep Brain Stimulation?
- Deep Brain Stimulation Procedures
- Recovering From A Deep Brain Stimulation Procedure
- Deep Brain Stimulation For Depression
- Risks Of Deep Brain Stimulation
- Transcranial Magnetic Stimulation VS Deep Brain Stimulation
- DBS vs TMS Comparison Table
- Types Of TMS
- Conclusion
What Is Deep Brain Stimulation?
Deep brain stimulation is a treatment that involves sending continuous electrical currents to specific areas of the brain to regulate the organ’s electrochemical activity and, therefore, the patient’s mood.
Deep Brain Stimulation Procedures
Deep brain stimulation requires an invasive medical procedure to implant small electrodes (a material that conducts electricity) in the brain (2, 3, 4, 5).
It is a multi-step procedure that follows this process:
- The patient’s suitability for DBS is assessed via thorough testing, usually involving an MRI or CT scan.
- An operation is carried out, usually while the patient is awake. The electrodes are introduced and carefully placed in the brain through tiny holes made in the skull.
- About one week later, an electrical pulse generator is implanted under the skin, near either the collarbone or the chest.
- The electrodes and the implantable pulse generator (IPG) are then connected with thin, insulated wires that are passed under the skin from the head, down the neck and shoulder, to the device.
- Once connected, the impulse generator continuously sends electrical currents through the electrodes to the brain.
- For the first four to six months, the doctor customizes the stimulation to the patient’s needs.
- Following the settings adjusted by the doctor, the patient controls the stimulator using a handheld remote. The doctor may suggest 24-hour stimulation or recommend the device be turned off at night during sleep.
Note that the procedure to implant a deep brain stimulation device is fully reversible.
Recovering From A Deep Brain Stimulation Procedure
After surgery, full recovery time is generally four to six weeks before heavy or strenuous activity, such as jogging or swimming, is permitted. The device is activated a few weeks after surgery and remains implanted for as long as the treatment is deemed necessary, which could be years.
Deep Brain Stimulation For Depression
Deep brain stimulation is approved by the FDA (Food and Drug Administration) for treating various disorders, including medically refractory epilepsy, Parkinson’s disease, and obsessive-compulsive disorder (OCD) (6,7,8).
Currently, the use of deep brain stimulation is still being studied in clinical trials for its use in treating major depression. It is only offered to patients on an experimental basis.
Results in trials are promising, with DBS consistently alleviating depression symptoms. However, more studies with larger population samples are needed before DBS becomes a viable mainstream treatment available for depression patients (9,10).
Risks Of Deep Brain Stimulation
While DBS can provide significant therapeutic benefits, it is not without risks, such as:
- Surgical risks: Because DBS is an invasive procedure, it carries minor risks of infection, internal bleeding, and complications related to anesthesia. Postoperative seizures can also occur in a small percentage of patients. Although relatively rare, they are a potential risk during the DBS implantation procedure.
- Hardware-related complications: The implanted electrodes or leads may shift from their intended position or even break, potentially leading to reduced efficacy or increased side effects. Device failure can necessitate additional surgery for replacement or repair.
- Battery depletion: The battery life of the implanted pulse generator (IPG) can vary based on usage and settings, typically lasting 3-5 years. When the battery depletes, surgical replacement is necessary, which carries additional surgical risks.
- Side effects: In rare cases, DBS can cause changes in mood, behavior, or cognitive function. These effects can include depression, anxiety, mania, impulsivity, apathy, and even suicidality.
- Personality changes: In rare cases, DBS can cause or exacerbate impulsive behaviors, hypersexuality, or even gambling tendencies, particularly when the electrodes are placed in the subthalamic nucleus.
A systematic review of studies concerning the use of DBS therapy has found a 16.7% complication rate overall, counting risks related to hardware, surgery, and neurological side effects (11).
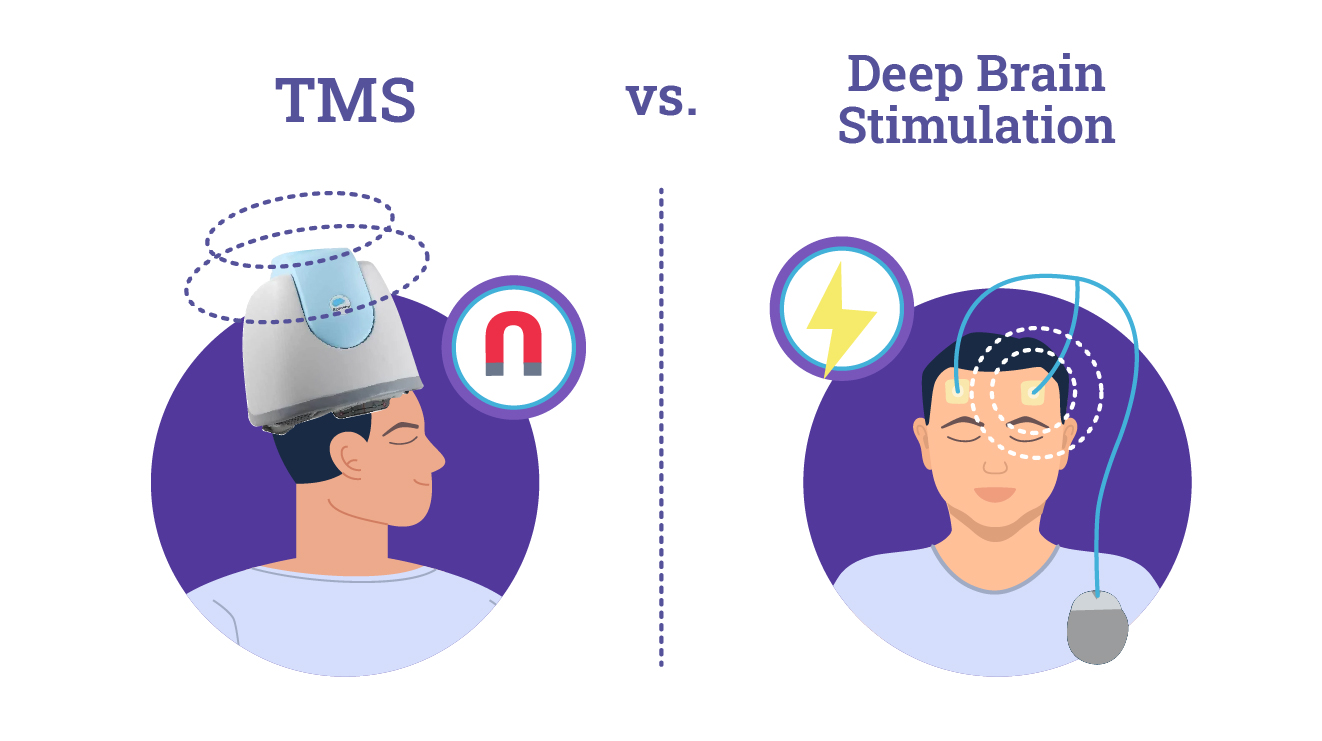
How Does Transcranial Magnetic Stimulation Differ From Deep Brain Stimulation?
How TMS Works: The “Depression Helmet”
In contrast to DBS, which requires surgery, TMS is a noninvasive procedure that can be performed at a doctor’s office without any anesthesia.
- During TMS, a head-mounted medical device containing electrical coils, sometimes called a “depression helmet,” is placed just above the patient’s scalp.
- An electrical current is passed through the coils, creating a magnetic field.
- This magnetic field stimulates deep neural networks in the targeted brain area, which in the case of depression is usually the dorsolateral prefrontal cortex.
- Neural stimulation balances the brain’s electrochemical activity, resulting in a more stable mood and reduced symptoms.
- The magnetic field induced during TMS is continuous and of the same strength throughout the session, which lasts roughly 20 minutes.
Is TMS Better Than DBS For My Condition?
Unlike DBS, TMS is FDA-approved for the treatment of depression, and studies show that it has a success rate of around 75% when it comes to relieving patients’ depression symptoms, with results lasting at least a year after treatment for many patients (12). TMS is also used for other disorders, such as anxiety and OCD. The FDA has approved it for these purposes since 2008 and 2009, respectively, and is reviewing the efficacy of TMS for ADHD.
In contrast to TMS, DBS is still not approved by the FDA for the treatment of depression. However, it is a well-established therapy for people with movement conditions and is FDA-approved for Parkinson’s disease, essential tremor, and dystonia. It is also used for psychiatric conditions such as OCD and epilepsy.
DBS vs TMS Comparison Table
| Comparison | TMS | DBS |
| Method | TMS is noninvasive and uses a helmet with electromagnetic coils to induce focused brain stimulation through the scalp, modulating neural activity. | DBS involves surgically implanting electrodes into specific brain regions and delivering continuous electrical pulses to regulate neural circuits. |
| Indications | Primarily used for treatment-resistant depression, anxiety disorders, OCD, and some neurological conditions like migraines. | A broad range of disorders, including Parkinson’s disease, essential tremor, OCD, and epilepsy. |
| Duration of Treatment | TMS typically involves daily sessions lasting 20-40 minutes over several weeks. | DBS is a long-term therapy, with continuous stimulation, often lasting years, if not indefinitely. |
| Side Effects | Generally mild, including scalp discomfort and headaches, and well tolerated by patients. TMS Seizures are rare (less than 1% of patients). | May cause surgical complications, infections, mood changes, cognitive issues, and hardware-related problems. |
| Cost | TMS is less expensive than DBS, with lower upfront costs of around $7,000-$8,000 for a complete course, but some patients may need to pay for repeated sessions. TMS is often covered by insurance. | DBS is more costly initially due to surgery and device implantation but may have lower long-term costs as maintenance is less frequent. Average costs, including surgery and maintenance, can range from $100,000-$150,000. As an experimental treatment, DBS is unlikely to be covered by insurance for treating depression. |
| Efficacy | Effective in treating depression OCD, anxiety, PTSD, and other conditions. | Can provide significant and long-lasting symptom relief for many neurological disorders |
| Contraindications | Metallic implants in the head, a history of seizures, or certain neurological conditions. | Any active infections, head implants, or significant cognitive impairment. |
Types Of TMS
Over the years, several types of TMS have been developed, each with unique protocols, equipment, and treatment paradigms. These include:
- Repetitive TMS (rTMS) involves delivering magnetic pulses at specific frequencies to stimulate brain regions like the dorsolateral prefrontal cortex (DLPFC) to treat depression. Considered the most typical form of TMS, it uses a figure-8 coil for precise targeting but is limited to stimulating superficial cortical areas.
- Deep TMS (dTMS) uses an H-coil to reach deeper brain regions involved in mood regulation, like the anterior cingulate cortex and insula. It is effective for treatment-resistant depression and OCD, offering broader stimulation than rTMS.
- Theta Burst Stimulation (TBS) is a kind of Deep TMS that delivers rapid bursts of high-frequency stimulation in a theta rhythm and comes in two types: intermittent (iTBS) and continuous (cTBS). TBS sessions are shorter (3-10 minutes) than standard rTMS and provide similar or better results for depression treatment.
- Bilateral TMS involves stimulating both hemispheres of the brain, usually the left and right DLPFC, in one session to balance neural activity. It may benefit patients who do not respond to unilateral TMS but requires longer sessions.
- Navigated TMS (nTMS) uses MRI-based neuronavigation to accurately target specific brain regions during treatment. This personalized approach can improve treatment outcomes by considering individual brain anatomy for precise stimulation.
Key Distinctions Between Types of TMS
| Type of TMS | Stimulation Depth | Treatment Duration | Efficacy | Applications |
| rTMS | Superficial cortical areas | 20-40 minutes | High for MDD | Major Depressive Disorder (MDD) |
| TBS (iTBS/cTBS) | Superficial cortical areas | 3-10 minutes | Similar to rTMS | MDD, efficient for short sessions |
| dTMS | Deeper subcortical areas | 20 minutes | Effective for treatment-resistant cases | MDD, OCD |
| Bilateral TMS | Both hemispheres (DLPFC) | 30-45 minutes | Potentially higher for non-responders | MDD, specific mood disorders |
| nTMS | Highly specific cortical areas | 20-40 minutes | Potentially higher accuracy | MDD, personalized for unique brain anatomy |
At Madison Avenue TMS & Psychiatry, we provide TMS therapy personalized to patients’ needs, using the latest approved innovations such as dTMS and TBS to maximize its effectiveness in reducing symptoms of depression and other mood disorders.
Conclusion
TMS and DBS are both effective and well-tolerated brain stimulation therapies with a solid foundation in evidence-based medicine. Both can help people with mental health struggles who do not respond well to conventional treatments. However, they have some important differences that should be considered before choosing one over the other.
As a noninvasive treatment, TMS has fewer risks and complications than DBS while still having its efficacy backed up by scientific studies and FDA approval. It is generally recommended for patients whose primary issue is with treatment-resistant depression, OCD, or anxiety and who want to have minimal disruption in their daily life, without the risks and recovery period associated with surgery. DBS, on the other hand, would be the better option for long-lasting neurological conditions such as Parkinson’s disease. Neither TMS nor DBS should be confused with electroconvulsive therapy (ECT), which is usually a second-line treatment for severely acute mental health conditions.
If you are interested in learning more about TMS or DBS therapy for depression or related mood disorders, you should consult with your doctor or a mental health professional who can provide you with more information and guidance on whether these treatments are suitable for you.
Madison Avenue TMS & Psychiatry offers psychotherapy, medication management services, and TMS for treating depression, anxiety, and other mental health conditions. Contact us online or call (212) 731-2033 if you have any questions or to make an appointment.
More resources on neurostimulation therapies:
Sources:
- Major depression. National Institute of Mental Health. Updated July 2023. Link. Accessed September 11, 2024.
- Deep brain stimulation. American Association of Neurological Surgeons. Link. Accessed September 11, 2024.
- Deep Brain Stimulation. Cleveland Clinic. Publication Date Unknown. Updated May 23, 2022. Link. Accessed September 11, 2024.
- Deep brain stimulation. Mayo Clinic. Published September 3, 2021. Link. Accessed September 11, 2024.
- Deep brain stimulation (DBS). Mayfield Brain & Spine. Updated April 2022. Link. Accessed September 11, 2024.
- FDA approval: medtronic deep brain stimulation for medically refractory epilepsy. Epilepsy Foundation. Published May 01, 2018. Link. Accessed May 6, 2022.
- Vercise deep brain stimulation (DBS) system – P150031. U.S. Food and Drug Administration. Publicaton Date Unknown. Updated January 27, 2021. Link. Accessed September 11, 2024.
- Deep brain stimulation: A way to rebalance neural circuits. International Neuromodulation Society. Published July 06, 2018. Updated December 13, 2019. Link. Accessed May 6, 2022.
- Remore, L. G., Tolossa, M., Wei, W., Karnib, M., Tsolaki, E., Rifi, Z., & Bari, A. A. Deep Brain Stimulation of the Medial Forebrain Bundle for Treatment-Resistant Depression: A Systematic Review Focused on the Long-Term Antidepressive Effect. Neuromodulation: Technology at the Neural Interface. 2024;27(4), 690-700. Link. Accessed September 11, 2024.
- Cha, J., Choi, K. S., Rajendra, J. K., McGrath, C. L., Holtzheimer, P. E., Figee, M., Kopell, B. H., & Mayberg, H. S. Whole brain network effects of subcallosal cingulate deep brain stimulation for treatment-resistant depression. Molecular Psychiatry. 2023; 29(1), 112-120. Link. Accessed September 11, 2024.
- Koh, Eun Jeong MD, PhD; Golubovsky, Joshua L BS; Rammo, Richard MD; Momin, Arbaz BS; Walter, Benjamin MD; Fernandez, Hubert H MD; Machado, Andre MD, PhD; Nagel, Sean J MD. Estimating the Risk of Deep Brain Stimulation in the Modern Era: 2008 to 2020. Operative Neurosurgery. 2021;21(5), p 277-290. Link. Accessed September 11, 2024.
- Deng, Y., Li, W., & Zhang, B. Functional Activity in the Effect of Transcranial Magnetic Stimulation Therapy for Patients with Depression: A Meta-Analysis. Journal of Personalized Medicine. 2023;13(3), 405. Link. Accessed September 11, 2024.
Dr. David Woo is the owner and head clinical psychiatrist at Madison Avenue TMS and Therapy in New York City. Dr. Woo has been seeing patients in private practice since 2002, always with the goals of combining evidence-based medicine with psychodynamic psychotherapy and collaborating with other mental health professionals to ensure the best possible outcomes for his patients. He has been certified to administer TMS at his practice since 2017. His greatest clinical interests include helping patients suffering from depression, anxiety, and obsessive compulsive disorder.
Follow Dr. Woo On
Dr. Woo On The Web



