What Is TMS Therapy?
TMS (transcranial magnetic stimulation) is a non-invasive treatment for major depression (MDD), obsessive-compulsive disorder (OCD), anxiety, attention-deficit hyperactivity disorder (ADHD), and many other conditions. During TMS treatment, a head-mounted coil, sometimes nicknamed the “depression helmet” is placed over the patient’s head. The doctor controls the coil as it sends subtle, barely perceptible magnetic currents through the scalp and skull to activate neurons in brain areas related to mood regulation.
Madison Avenue TMS & Psychiatry offers TMS, as well as traditional treatments like talk therapy and medication management, to patients in the New York City area. Let’s explore how TMS works, its benefits and risks, and whether TMS might be right for you.
- Introduction To TMS
- How Does TMS Work In The Brain To Treat Depression?
- Conditions That Transcranial Magnetic Stimulation (TMS) Can Treat
- Who Can Benefit From TMS
- How Effective Is TMS Treatment?
- What To Expect from TMS Therapy, From Start To Finish
- Types Of TMS
- TMS Myths And Facts
- Frequently Asked Questions On TMS Treatment
Introduction To TMS
TMS is an innovative therapy that is most often prescribed as a treatment for depression and OCD patients who have not found relief from their symptoms using traditional medications such as antidepressants.
Many symptoms of depression and other mental health conditions are caused by underactive neurons (nerve cells) that cause imbalances in your natural brain chemistry. TMS therapy stimulates these underactive neurons, restoring normal activity and helping to relieve or eliminate symptoms.
TMS works best as a targeted therapy, with the pulses directed precisely to the brain area related to the patient’s symptoms. Before any pulses are administered, TMS therapy requires extensive mapping of a patient’s brain to target where the electric currents should be applied for best results.
30%
30% + of individuals who have depression don’t benefit from antidepressants (1).
Since 2008
In 2008, TMS was approved by the FDA for treatment-resistant depression.
20 million
Over 20 million TMS treatments are estimated to have been delivered in the US.
in 19 minutes
The average TMS session is delivered in 19 minutes.
The US Food and Drug Administration (FDA) authorized TMS as a treatment for depression in 2008. It is an outpatient treatment administered without anesthesia, allowing patients to return to regular activity directly following treatment. Side effects reported after TMS are rare and usually mild, with most patients only experiencing minor discomfort, e.g., mild headache or scalp irritation, easily treated with over-the-counter meds.
Patients undergoing TMS are not required to participate in any additional medication regimen. However, it is safe to take TMS with pharmaceutical medications. In fact, this kind of combination therapy, if monitored by your psychiatrist, can even lead to improved results from both treatments.
At Madison Avenue TMS & Psychiatry, your care is our highest priority. We are dedicated to providing you with all the necessary tools and information to make your TMS therapy as comfortable and effective as possible.
Our friendly staff is available to address any of your questions or concerns every step of the way. They will help you understand how TMS is used in the treatment of depression, what to expect from TMS therapy, and support you in your mental health journey.
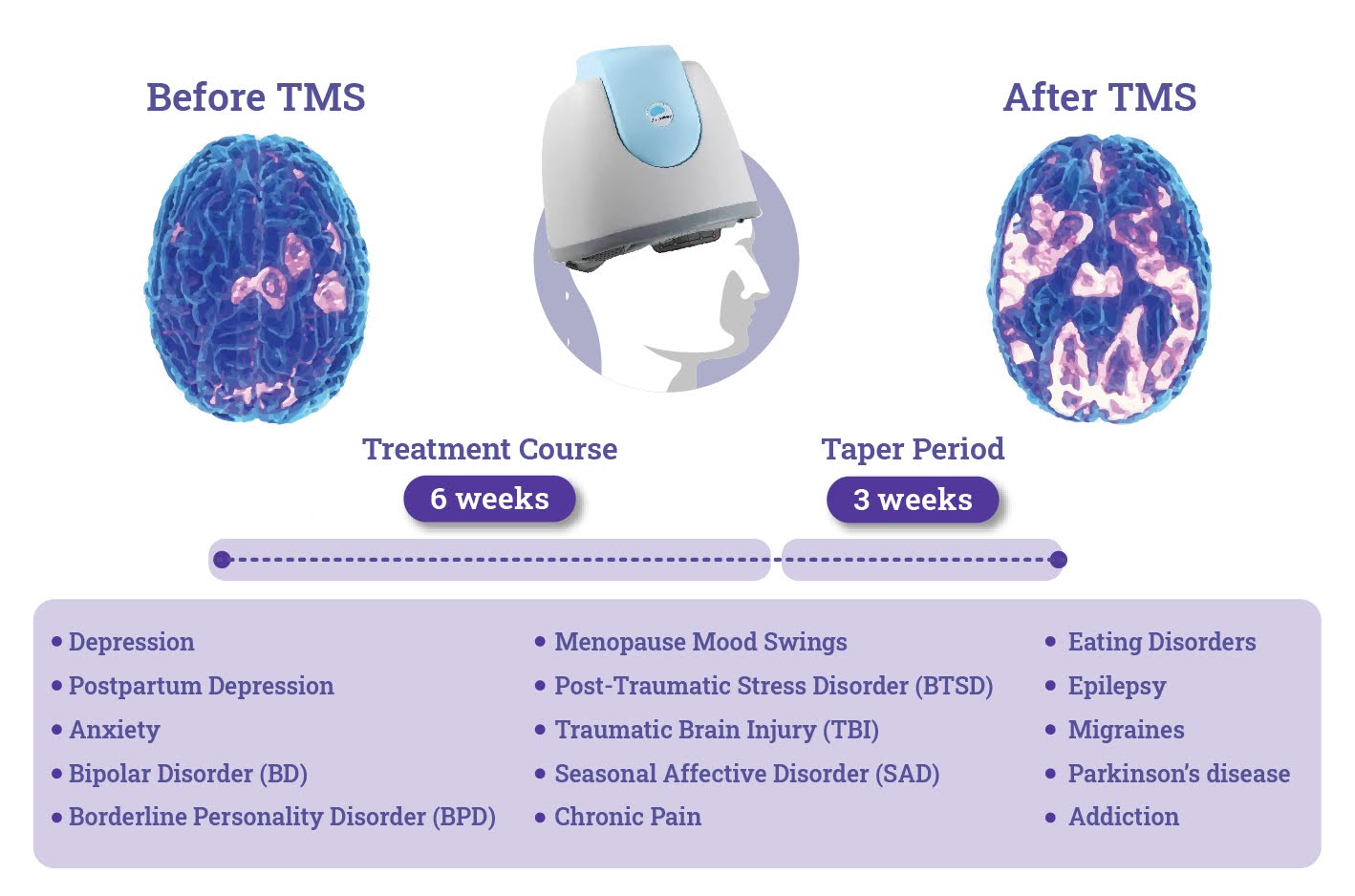
Conditions that Transcranial Magnetic Stimulation (TMS) Can Treat
In the United States, TMS is only FDA-approved for MDD and OCD. However, many clinical trials have shown that TMS can safely and effectively treat other conditions, such as:
- Epilepsy
- Menopause mood swings
- Migraines
- Parkinson’s disease
- Postpartum Depression
- Post-Traumatic Stress Disorder (PTSD)
- Seasonal affective disorder (SAD)
- Traumatic brain injury (TBI)
Learn more about other conditions TMS can treat.
Who Can Benefit from TMS
As a drug-free depression treatment, TMS is recommended for patients who:
- Have tried various rounds of different medications and psychotherapy without success
- Want to avoid the side effects of medication (such as sexual dysfunction and weight gain)
- Are pregnant or planning to become pregnant
- Do not have a seizure disorder or a history of seizures
- Do not have any metal implants inside the head
How Effective Is TMS Treatment?
What to Expect from TMS Therapy, from Start to Finish
Step 1: Preliminary Phone Screening
If you’re interested in finding out whether TMS treatment is right for you, please take our self-assessment.
If you’d like to commence treatment, please contact us for a preliminary phone screening.
During this initial conversation, we will gather some basic information, ask about your insurance coverage, and answer any questions you may have about undergoing TMS therapy. Once we’ve checked with your insurance company, we will be back in touch to discuss insurance coverage/payment options.
Step 2: Pre-Treatment Evaluation
The initial consultation is an opportunity for Dr. David Woo to get to know you, your symptoms, and your medical history to determine if TMS therapy is the right treatment option for you and to tell you more about the TMS treatment process. Following this appointment, we will coordinate your insurance coverage with your provider. We can also work with you to arrange an out-of-pocket payment plan if necessary.
Step 3: Brain Mapping + First Treatment
The first step for successful TMS treatment is effectively mapping the patient’s brain to determine which point on the head should receive stimulation. During the mapping session, the technician will use the TMS device to send magnetic pulses and register the brain’s response until the optimal point is identified. This step takes approximately 45 minutes, including the mapping process and the initial TMS treatment.
Step 4: 6-Week Treatment Course
Patients come in for treatment five days per week (Monday-Friday) for six weeks. After mapping, subsequent treatment sessions last approximately 20 minutes. Our treatment rooms are designed to be comfortable, and we invite you to relax, watch TV, or listen to your choice of music during sessions. Patients are asked to complete a weekly evaluation to measure progress. If determined necessary at any point during treatment, the technician may perform a re-mapping to improve treatment effectiveness.
3-Week Taper Period
After the six-week treatment course, there is a three-week taper period. Patients will need to come into the office for three TMS sessions in the seventh week, two sessions in the eighth week, and one final session in the ninth week.
Types Of TMS
Several types of TMS have emerged since the treatment was first developed, each with unique protocols, equipment, and approaches to treatment. These include:
- Deep TMS (dTMS) Uses an H-coil (like the BrainsWay device) to reach deeper brain regions, such as the anterior cingulate cortex and insula. It is highly effective for treatment-resistant depression and OCD and offers a more targeted form of stimulation than rTMS. This is the type of TMS that we specialize in at Madison Avenue TMS & Psychiatry.
- Theta Burst Stimulation (TBS) is a variant of Deep TMS that delivers rapid bursts of high-frequency stimulation in a theta rhythm (one of the natural brain wave patterns). TBS Sessions are usually shorter (3-10 minutes) than typical rTMS treatment and provide similar or better results when it comes to relieving depression symptoms.
- Repetitive TMS (rTMS) is the most typical, “standard” form of TMS. It delivers magnetic pulses at particular frequencies to stimulate brain regions to treat depression. rTMS uses a figure-8 coil for precise targeting but cannot penetrate the brain as deeply as other methods.
- Bilateral TMS is TMS applied to both hemispheres of the brain, usually the left and right DLPFC, which aims to balance activity on both sides. Bilateral TMS takes longer than regular TMS, but it can be a good option for patients who are unresponsive to unilateral TMS.
- Navigated TMS (nTMS) is TMS therapy combined with MRI-based neuronavigation to help the doctor target specific brain regions more accurately.
At Madison Avenue TMS & Psychiatry, we provide TMS therapy personalized to patients’ needs, using the latest approved innovations such as dTMS and TBS to maximize its effectiveness in reducing symptoms of depression and other mood disorders.
Different Types of TMS Compared
| Type of TMS | Stimulation Depth | Session Duration | Benefits | Approved Applications |
|---|---|---|---|---|
| rTMS | Superficial brain areas | 20-40 minutes | High for MDD | Major Depressive Disorder (MDD) |
| TBS | Superficial brain areas | 3-10 minutes | Similar to rTMS, shorter sessions | MDD |
| dTMS | Deeper brain areas | 20 minutes | Especially effective for treatment-resistant cases | MDD, OCD |
| Bilateral TMS | Both hemispheres (DLPFC) | 30-45 minutes | Potential benefits for non-responders | MDD, specific mood disorders |
| nTMS | Highly targeted brain areas | 20-40 minutes | Potentially more accurate | MDD, personalized for unique brain anatomy |
TMS Myths And Facts
Myth: TMS Isn’t Safe
Fact: Repeated medical studies and multiple meta-analyses support TMS’s safety and effectiveness (5). The FDA approved it in October 2008, and the medical community widely considers it a very low-risk procedure, as TMS does not require anesthesia and can be performed as an outpatient procedure.
Most patients only report minor discomfort as a side effect and can continue their life as usual directly following each procedure.
Other reported side effects of TMS treatment include:
- Difficulty sleeping
- Eye pain
- Facial pain/toothache
- Muscle twitch
In very rare cases (0.1%), patients may experience seizures after treatment. However, it is important to note that no adverse cognitive effects, including memory loss, have been reported following TMS.
Patients and caregivers should be vigilant of behavioral changes during treatment — especially if they notice an increase in suicidal thoughts or behavior or if depression symptoms worsen.
Overall, the side effects of TMS treatment are few and low in severity when compared to the commonly reported side effects of antidepressant medications. TMS provides a safe and effective alternative to these treatment methods. Learn more about TMS therapy success rates.
Myth: Nothing Has Worked For Me, So TMS Won’t Work Either
Fact: TMS is recommended for patients who have tried many other therapies— including multiple medications and psychotherapy —with no relief. Clinical data for over 1000 participants undergoing a course of deep TMS treatment for MDD, some 75% of participants achieved a clinical response, with one out of two achieving remission.
Myth: TMS Isn’t Covered By Insurance
Fact: Nearly all major insurance providers offer coverage for TMS. Even if we’re not in-network with your insurance plan, we can negotiate coverage as an out-of-network provider in most cases.
Learn more about TMS and insurance here. You can also learn more about TMS therapy costs.
Frequently Asked Questions on TMS Treatment
1. Are TMS and ECT the same procedure?
TMS is not the same as electroconvulsive therapy (ECT, formerly called “electroshock therapy” or “shock treatment”). While both therapies are used to treat major depression, ECT is usually administered in a hospital with anesthesia, which adds to the risk and recovery time of the procedure. Most physicians recommend ECT as an emergency measure because it is invasive and can lead to serious side effects, like memory loss. TMS is a different form of brain stimulation. It’s a non-invasive outpatient procedure that has demonstrated few to no side effects.
2. What are the side effects of TMS?
Only a few (usually minor) side effects are reported by patients, and the majority of patients do not stop treatment due to them. The most common side effect is slight pain or discomfort in the treatment area, usually only reported in the first week. Learn more about the potential side effects of TMS.
3. What can I do and not do after a TMS session?
You can return to your normal daily activities immediately after each TMS session.
4. Can TMS be combined with other depression treatments, like my current medication?
TMS can be combined with most antidepressant medications, but Dr. Woo will evaluate all of your current medications and their compatibility during your pre-treatment evaluation appointment. You’re welcome to continue any psychotherapy you’re currently undergoing, or if you’re interested, we can offer you psychotherapy and medication management services.
5. When will I start to see results with TMS?
The time it takes to see results with TMS will vary from patient to patient. Some patients report an improvement in depression symptoms in as little as 2 weeks, while others don’t notice an improvement until week 4 or 5. Patients who take longer to notice symptom relief are still receiving effective treatment and can still experience long-lasting relief from their depression symptoms. Learn more about what can affect your time to results.
6. How long do TMS results last?
How long the effects of TMS last will vary from patient to patient. Studies show that patients who achieve remission from depression are more likely to continue to show signs of remission or response to treatment even one year after completing a round of treatment. In some cases, patients may need a maintenance session of TMS. Learn more about what influences how long TMS results can last.
7. Who will I interact with during treatment?
Throughout therapy at Madison Avenue TMS & Psychiatry, you will see a technician at each of your appointments who will guide you through each treatment. Dr. Woo will check in on your progress at least weekly, and he’s available for additional check-ins anytime you’d like.
8. Is TMS right for me?
TMS is considered when patients do not see improvement in depression symptoms after trying at least 2 types of antidepressants. TMS cannot be administered to patients with implanted metallic devices in or around the head (such as aneurysm clips or coils and electrodes that monitor brain activity) or electrical implants (such as pacemakers and ICDs).
Learn more about who is a good candidate for TMS.
Sources:
- Aditya Somani, Sujita Kumar Kar – Efficacy of repetitive transcranial magnetic stimulation in treatment-resistant depression: the evidence thus far. General Psychiatry. 2019;32:e100074. Link. Accessed September 24, 2024.
- Roth, Y., Hanlon, C. A., Pell, G., Zibman, S., Harmelech, T., Muir, O. S., MacMillan, C., Prestley, T., Purselle, D. C., Knightly, T., & Tendler, A. Real world efficacy and safety of various accelerated deep TMS protocols for major depression. Psychiatry Research. 2023;328, 115482. Link. Accessed September 24, 2024
- George MS et al. Daily Left Prefrontal Transcranial Magnetic Stimulation Therapy for Major Depressive Disorder. Arch Gen Psychiatry. 2010;67(5):507-16. Link. Accessed September 24, 2024.
- Dunner DL et al. A multisite, naturalistic, observational study of transcranial magnetic stimulation for patients with pharmacoresistant major depressive disorder: durability of benefit over a 1-year follow-up period. J Clin Psychiatry. 2014;75(12):1394-401. Link. Accessed September 24, 2024.
- Brini, S., Brudasca, N., Hodkinson, A., Kaluzinska, K., Wach, A., Storman, D., Prokop-Dorner, A., Jemioło, P., & Bala, M.. Efficacy and safety of transcranial magnetic stimulation for treating major depressive disorder: An umbrella review and re-analysis of published meta-analyses of randomised controlled trials. Clinical Psychology Review. 2023;100, 102236. Link. Accessed September 24, 2024.



